Research
|
Although 90% of cancer deaths are caused by metastasis, the underlying pathogenic mechanisms are poorly understood. Cell plasticity is the ability of cells to reversibly change their phenotype and plays a critical role at multiple steps in the complex process of tumour dissemination, which relies on cells undergoing an epithelial-to-mesenchymal transition (EMT), migrating away from the primary tumour and later undergoing a reverse mesenchymal-to-epithelial transition (MET). While several transcription factors that regulate EMTs have been isolated, how these actually impinge on the cellular responses underlying cell plasticity remains poorly understood. Even less is known about MET.
|
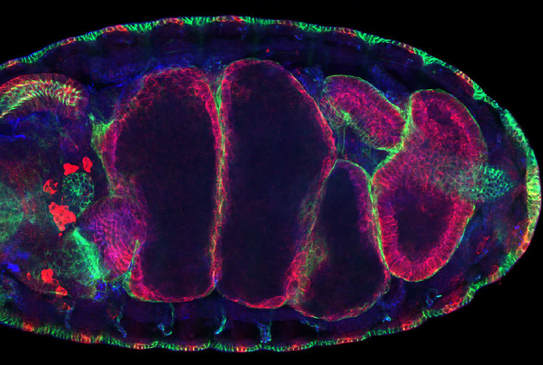
Cell Plasticity in DevelopmentThe embryonic Drosophila midgut offers an unparalleled opportunity to visualise these processes in vivo and delineate the transcriptional networks and downstream effectors involved. During morphogenesis, midgut cells undergo an EMT, enabling their collective migration through the embryo, and then a reverse MET, re-epithelializing to form the embryonic midgut epithelium. Experimentally, the midgut is highly accessible, amenable to genetic manipulation and we have developed methods to image the cells to a subcellular resolution and track them in 4D.
|
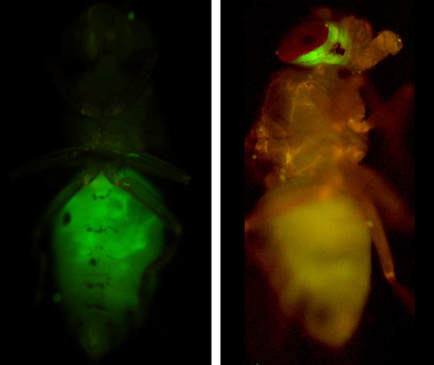
Cell Plasticity in Tumour ProgressionIn many cancers, transcription factors capable of inducing motile migratory cell behaviours, so-called EMT transcription factors, are found highly expressed. Moreover, the expression of these genes is not restricted to metastatic invasive cancers; they are often found in benign tumours, or in tumours long before they show any sign of metastasis. Our lab has been working on the development of model systems for studying the roles of EMT transcription factors in primary tumour growth; and in collaboration with the lab of Dr. Andreu Casali, the roles of epithelial cell plasticity in metastatic tumour progression.
|